Secret sauce that brings YouTube followers, views, likes
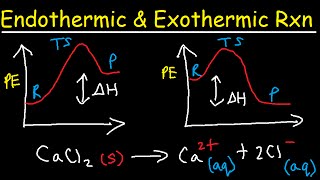
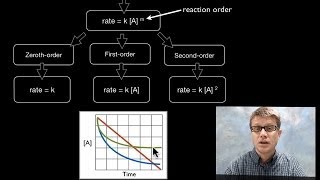
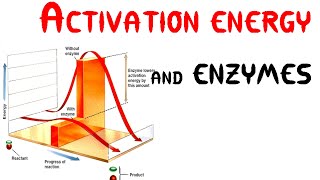
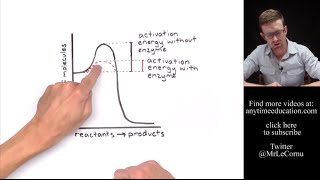
Activation Energy and Catalysts
Activation Energy is the minimum amount of energy required for a reaction to occur. Catalysts often help a reaction occur more quickly by lowering the activation energy requirement.
Helpful Links:
https://www.khanacademy.org/science/h...
https://www.khanacademy.org/science/c...
https://www.sciencenewsforstudents.or...
http://ch302.cm.utexas.edu/kinetics/c...
Check out my other social media!
Facebook @teachmetoscience
Instagram @teachmetoscience
Twitter @TeachMetoScien1
Website teachmetoscience.com
I absolutely love teaching. However, part of teaching is being a lifelong learner. So if you have any recommendations or ideas for how I can make Teach Me to Science even better, let me know by leaving a comment down below!
Recommended