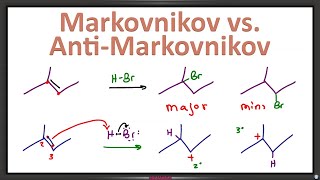
Anti-Markovnikov Radical Halogenation of Alkenes
https://Leah4sci.com/Radical presents: AntiMarkovnikov Radical Halogenation of Alkenes
Need help with Orgo? Download my free guide ’10 Secrets to Acing Organic Chemistry’ HERE: https://leah4sci.com/orgoebook/
In this video, you’ll learn how the radical halogenation of an alkene in the presence of peroxides differs from the typical hydrohalogenation alkene addition reaction. You’ll learn how to apply initiation and propagation steps of the radical mechanism, how to identify the most stable intermediate, and understand why the antiMarkovnikov product is favored.
You’ll also learn about the stereochemistry of alkene radical halogenation.
Links & Resources Mentioned In This Video:
https://Leah4sci.com/AlkeneReactions Alkene Reaction Mechanisms
https://Leah4sci.com/Markovnikov Markovnikov’s Rule in Alkene Addition Reactions
https://Leah4sci.com/Carbocation Carbocation Stability and Ranking
In this video:
[0:25] Definition of Radical Halogenation (The “Light” Reaction)
[2:53] Initiation Step with Alkyl Peroxide
[3:54] Propagation Step
[4:55] Continuation of Propagation with Alkene
[5:32] Stability of the Radical Intermediate
[7:18] Formation of Final Product
[7:55] Stereochemistry of Reaction
Watch Next: Cyclopropanation of Alkenes • Cyclopropanation of Alkenes Carbene v...
Catch the entire Radical Reactions Series on my website at http://Leah4sci.com/Radical
Follow along with your semester by using my Orgo Syllabus Companion: https://leah4sci.com/syllabus
For more indepth review including practice problems and explanations, come join my online membership site the organic chemistry study hall: https://leah4sci.com/join
For private online tutoring visit my website: https://leah4sci.com/organicchemistry
For questions and comments, find me on social media here:
Facebook: / leah4sci
Twitter: / leah4sci
Instagram: / leah4sci
Pinterest: / leah4sci