Electrolysis of Water - Electrochemistry
This chemistry video tutorial provides a basic introduction into the electrolysis of water which splits H2O into H2 (hydrogen has) and O2 (oxygen gas). Oxygen gas is produced at the anode and hydrogen gas is produced at the cathode. Electrons flow from the anode to the cathode. Oxidation always occurs at the anode and reduction occurs at the cathode.
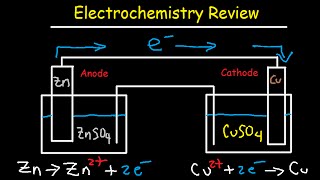
Intro to Galvanic & Voltaic Cells:
• Introduction to Galvanic Cells & Volt...
How To Draw Galvanic Cells:
• How To Draw Galvanic Cells and Voltai...
Standard Reduction Potentials:
• Standard Reduction Potentials of Half...
Cell Potential Problems:
• Cell Potential Problems Electrochem...
Cell Notation Problems:
• Cell Notation Practice Problems, Volt...
___________________________________
Concentration Cells:
• Concentration Cells & Cell Potential ...
Cell Potential & Gibbs Free Energy:
• Cell Potential & Gibbs Free Energy, S...
Cell Potential & Equilibrium K:
• Equilibrium Constant K & Cell Potenti...
Nernst Equation:
• Nernst Equation Explained, Electroche...
Electrolysis of Water:
• Electrolysis of Water Electrochemistry
_____________________________________
Electrolysis of Sodium Chloride:
• Electrolysis of Sodium Chloride Ele...
Electrolysis & Electroplating Problems:
• Electrolysis & Electroplating Practic...
Electrochemistry Practice Problems:
• Electrochemistry Practice Problems ...
SAT Chemistry Subject Test Review:
• SAT Chemistry Subject Test
Carbon 14 Dating:
• Carbon 14 Dating Problems Nuclear ...
Beer Lambert's Law:
• Beer Lambert's Law, Absorbance & Tran...
______________________________________
Final Exams and Video Playlists:
https://www.videotutor.net/
FullLength Videos and Worksheets:
/ collections
Chemistry PDF Worksheets:
https://www.videotutor.net/chemistry...