A little secret to rock your YouTube subscribers
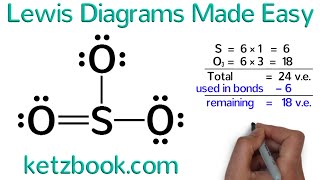
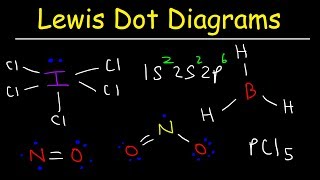
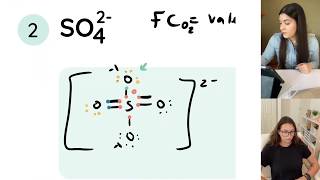
Lewis Structure of Formic Acid (HCOOH or CH2O2)
Formic Acid is a carboxylic acid with a single H as a substituent on the carbon. The carbon is DOUBLEbonded to one of the oxygens, and then the OTHER oxygen has the extra H on it.
Check me out: http://www.chemistnate.com
Recommended