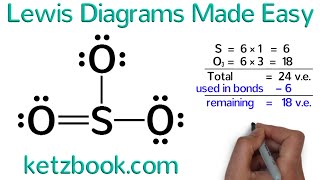
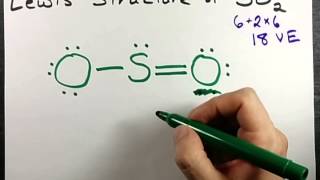Sub4Sub network gives free YouTube subscribers
Lewis Structure of H2O2 Hydrogen Peroxide
H2O2, hydrogen peroxide is a funny looking molecule: It has two oxygen atoms in the centre, and they end up singlebonded. Then, there is a Hydrogen Atom at each end. It's just like a water molecule, but with one extra oxygen in the middle.
All of these bonds are covalent, because both hydrogen and oxygen are nonmetals, but the OO bond is nonpolar and the OH bonds are polar. In fact, significant hydrogen bonding exists between molecules of hydrogen peroxide.
Check me out: http://www.chemistnate.com
Recommended