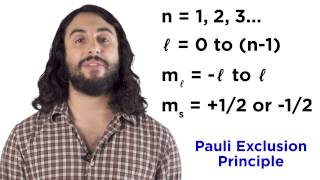Orbitals: Crash Course Chemistry #25
In this episode of Crash Course Chemistry, Hank discusses what molecules actually look like and why, some quantummechanical threedimensional wave functions are explored, he touches on hybridization, and delves into sigma and pi bonds.
Pssst... we made flashcards to help you review the content in this episode! Find them on the free Crash Course App!
Download it here for Apple Devices: https://apple.co/3d4eyZo
Download it here for Android Devices: https://bit.ly/2SrDulJ
Table of Contents
Molecules: Clumpy Globs... 0:18
QuantumMechanical ThreeDimensional Wave Functions 3:06
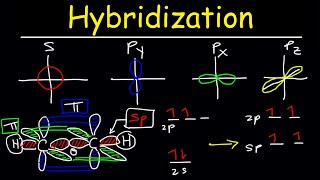
S & P Orbital Hybridization 5:27
Sigma & Pi Bonds 7:32
Hybridized Orbitals 5:52
Crash Course is on Patreon! You can support us directly by signing up at / crashcourse
Want to find Crash Course elsewhere on the internet?
Facebook / youtubecrashcourse
Twitter / thecrashcourse
Instagram / thecrashcourse
CC Kids: / crashcoursekids