How to get free YouTube subscribers, likes and views?
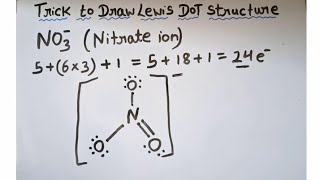
Resonance Structures of NO3(-1) nitrate ion
There are three equallyvalid Lewis structures for the nitrate ion, which is one nitrogen atom surrounded by three oxygen atoms (with one bonus electron from the 1 charge). These combine to make a "resonance hybrid" which reflects the delocalized electrons among the three NO bonds.
Check me out: http://www.chemistnate.com
Recommended